
BIGBEAR
PHARMACEUTICALS
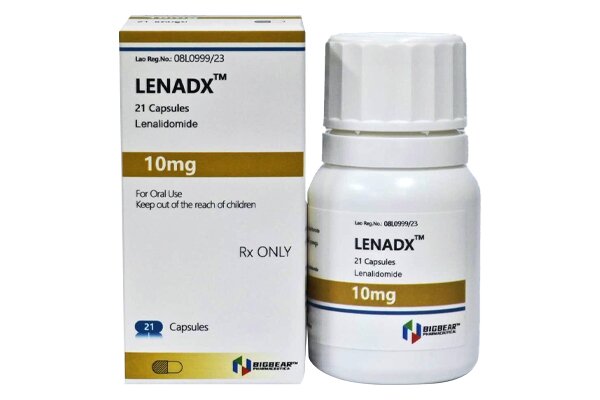

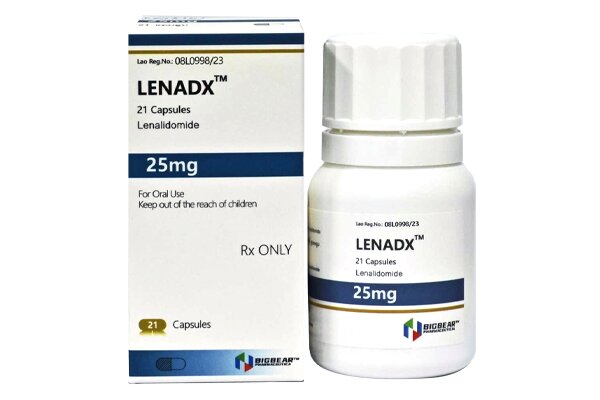
Lenalidomide is an immunomodulatory agent and a thalidomide analogue with anti-angiogenic and anti-tumor properties.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Regulatory Expansion into Frontline TherapyOn February 18, 2015, Celgene Corporation announced that the U.S. Food and Drug Administration (FDA) has ex···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 73
Regulatory Approval for Advanced Mantle Cell LymphomaOn June 5, 2013, Celgene Corporation announced that the U.S. Food and Drug Administration (FDA) a···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 72
Expanded Indication for Darzalex in Relapsed or Refractory Multiple MyelomaOn November 21, 2016, Janssen Biotech, Inc. announced that the U.S. Food an···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 73
Strategic Expansion of Revlimid Maintenance TherapyOn February 22, 2017, Celgene Corporation announced that the U.S. Food and Drug Administration (FDA···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 72
Overview of EltrombopagEltrombopag is an oral non-peptide thrombopoietin receptor agonist (TPO-RA). It increases platelet production by activating the···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 73
Regulatory Milestone and Expanded Treatment IndicationOn June 27, 2019, the Janssen Pharmaceutical Companies of Johnson & Johnson announced that t···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 71
Regulatory Milestone for Pfizer’s Oncology PortfolioOn February 12, 2025, Pfizer Inc. (NYSE: PFE) announced that the U.S. Food and Drug Administration···【Read More】
Update: 19 Mar,2026Source: Haiou HealthViews: 74
Copyright2024@ BIGBEAR All right reserved BIGBEAR



